Analytical Chemistry Final- ACS Exam
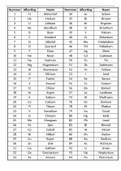
ppm - (grams analyte/grams sample)x10^6 Molarity - moles analyte/liter of solution Volume Percent - (volume solute/volume soution)x100 Volume ppm - (volume solute/volume solution)x10^6 kilo- - 10^3 deci- - 10^-1 centi- - 10^-2 milli- - 10^-3 micro- - 10^-6 nano- - 10^-9 pico- - 10^-12 femto- - 10^-15 weight percent - (grams analyte/grams sample)x100 ppt - (grams analyte/grams sample)x10^3 ppt simplified - gram analyte/liter solution ppm simplified - mg analyte/liter solution ppb simplified - micrograms analyte/liter solution pptr simplified - nanograms analyte/liter solution buoyancy correction - m=(m'(1-(air density/weight density)))/(1-(air density/object density)) accuracy - closeness of the mean to the "true value" precision - reproducibility of individual measurements Uncertainty in Addition/Subraction - e=sqrt(ex1^2+ex2^2+ex3^2+...) Uncertainty in Multiplication/Division - e=y*sqrt((ex1/x1)^2+(ex2/x2)^2+(ex3/x3)^2+...) Significant Figures in Logarithms and antilogarithms - the number of significant figures in the log should equal the number of digits in the mantissa How many significant figures in log(205.5) - four significant figures, so you will need four decimal places in your answer pH - -log[H3O+] [H3O+] - 10^-pH Absorbance - -log(transmittance) Random Error - -repeated measurements are sometimes high and sometimes low -cannot be corrected for Systematic Error - -repeated measurements are usually always high or always low -can and should be corrected for Relative uncertainty= - absolute uncertainty/magnitude of measurement 68% of measurements in a Gaussian Curve will lie - between the mean-1 and the mean+1 Variance in standard deviation - standard deviation squared mean= - true value +-time*standard deviation T-test Case 1 - measure sample of known composition T-test case 2 - compare replicate measurement of an unknown sample T-test case 3 - compare individual difference of an unknown sample - two sets of data analyzed by both methods being used T-test case 1 equation - true value= mean (+-) (time*standard deviation)/sqrt(number of measurements)) T-test case 1 Tcalc= - (sqrt(n)Iknown value-calculated meanI)/standard deviation For Case 1: If TcalcTtable - the actual value isn't in the range and it is bad
École, étude et sujet
- Établissement
- Analytical Chemistry
- Cours
- Analytical Chemistry
Infos sur le Document
- Publié le
- 15 novembre 2023
- Nombre de pages
- 12
- Écrit en
- 2023/2024
- Type
- Examen
- Contient
- Questions et réponses
Sujets
-
analytical chemistry final acs exam
Document également disponible en groupe